Weitere Formate
Autorenfreundlich Bücher kaufen?!
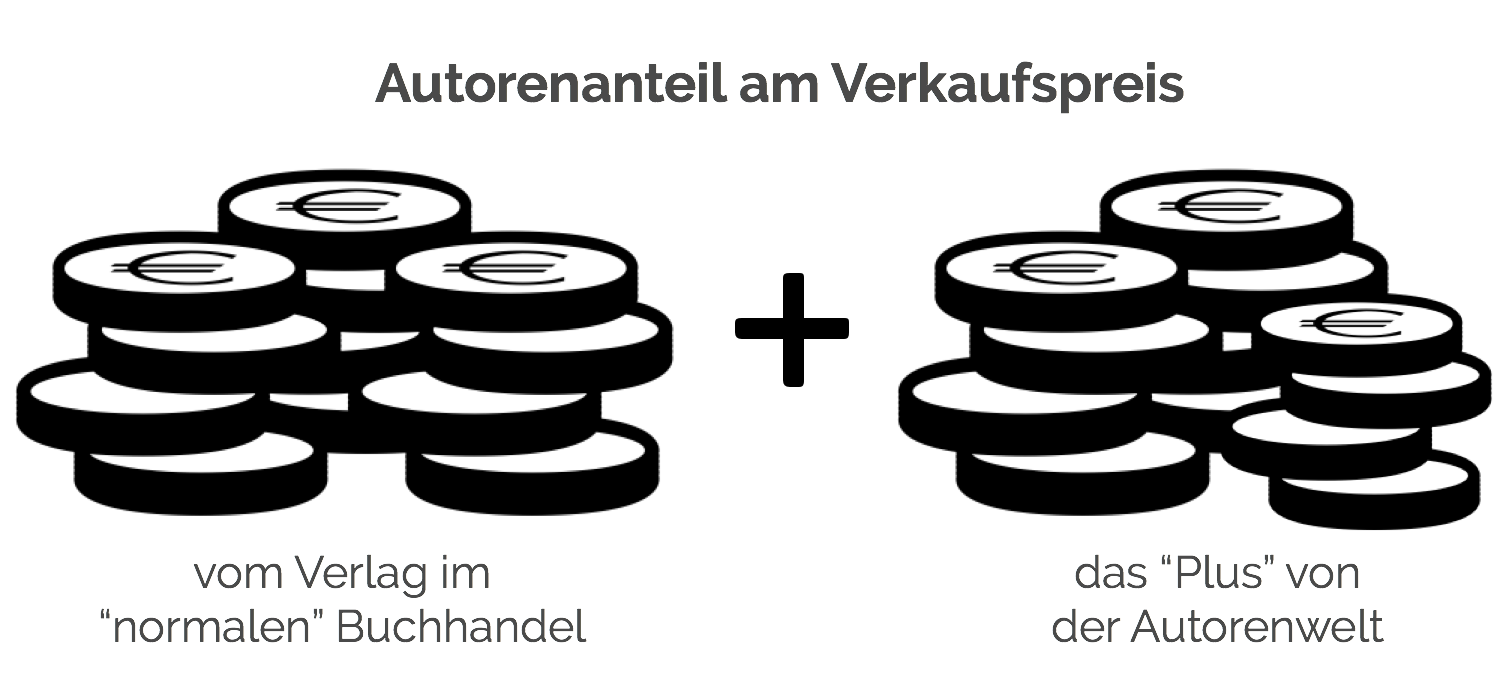
Beschreibung
Details
| Verlag | Springer Berlin |
| Ersterscheinung | März 2012 |
| Maße | 23.5 cm x 15.5 cm |
| Gewicht | 516 Gramm |
| Format | Softcover |
| ISBN-13 | 9783642261916 |
| Auflage | 2010 |
| Seiten | 320 |
✍️ 🧑🦱 💚 Autor:innen verdienen bei uns doppelt. Dank euch haben sie so schon 253.133 € mehr verdient. → Mehr erfahren 💪 📚 🙏

| Verlag | Springer Berlin |
| Ersterscheinung | März 2012 |
| Maße | 23.5 cm x 15.5 cm |
| Gewicht | 516 Gramm |
| Format | Softcover |
| ISBN-13 | 9783642261916 |
| Auflage | 2010 |
| Seiten | 320 |