Weitere Formate
Autorenfreundlich Bücher kaufen?!
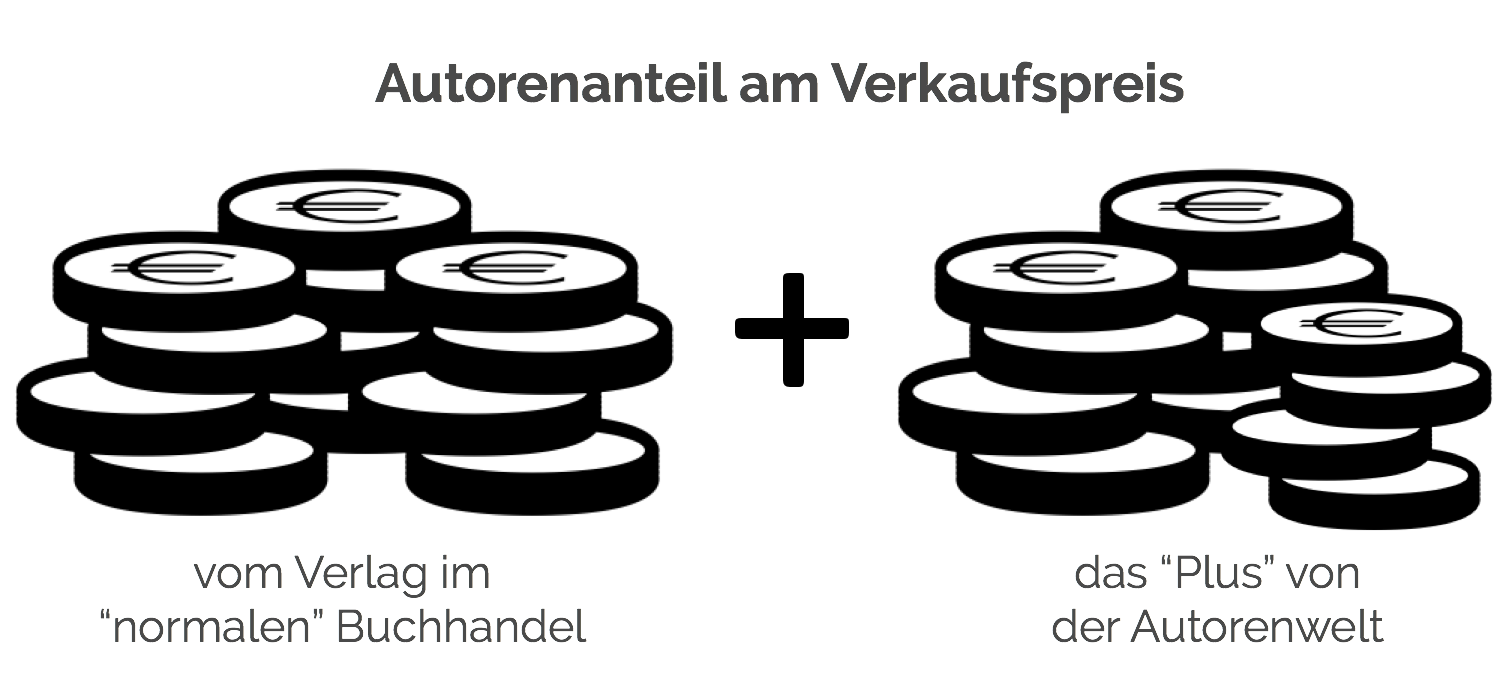
Beschreibung
Contributors: W. Kewenig; G. Forlani, J. Marriage, G. Ulloa
This book is based upon a report prepared at the request of the European Commission. Its purpose is to set out the existing laws regulating the development, production, distribution and use of in vitro diagnostic medical devices (IVDMD) in the U.K., Germany, France, Italy and Spain. Each national section has the same structure and covers the same ground in order to enable comparisons. IVDM Devices includes not only reagents, but also technical instruments, or combination of both, for human use. The book is directed towards industry and the regulation is covered with a high degree of specificity. This reference book, completely written in English, is the only comprehensive exposition of the laws of the five countries regarding IVDM Devices available. It is also the most convenient means of comparing those laws. As such, it will be a useful guide to all IVDM Devices manufacturers interested in the European market.
Details
| Verlag | Springer Netherland |
| Ersterscheinung | 01. Dezember 2010 |
| Maße | 24 cm x 16 cm |
| Gewicht | 213 Gramm |
| Format | Softcover |
| ISBN-13 | 9789048144396 |
| Seiten | 118 |


