Weitere Formate
Autorenfreundlich Bücher kaufen?!
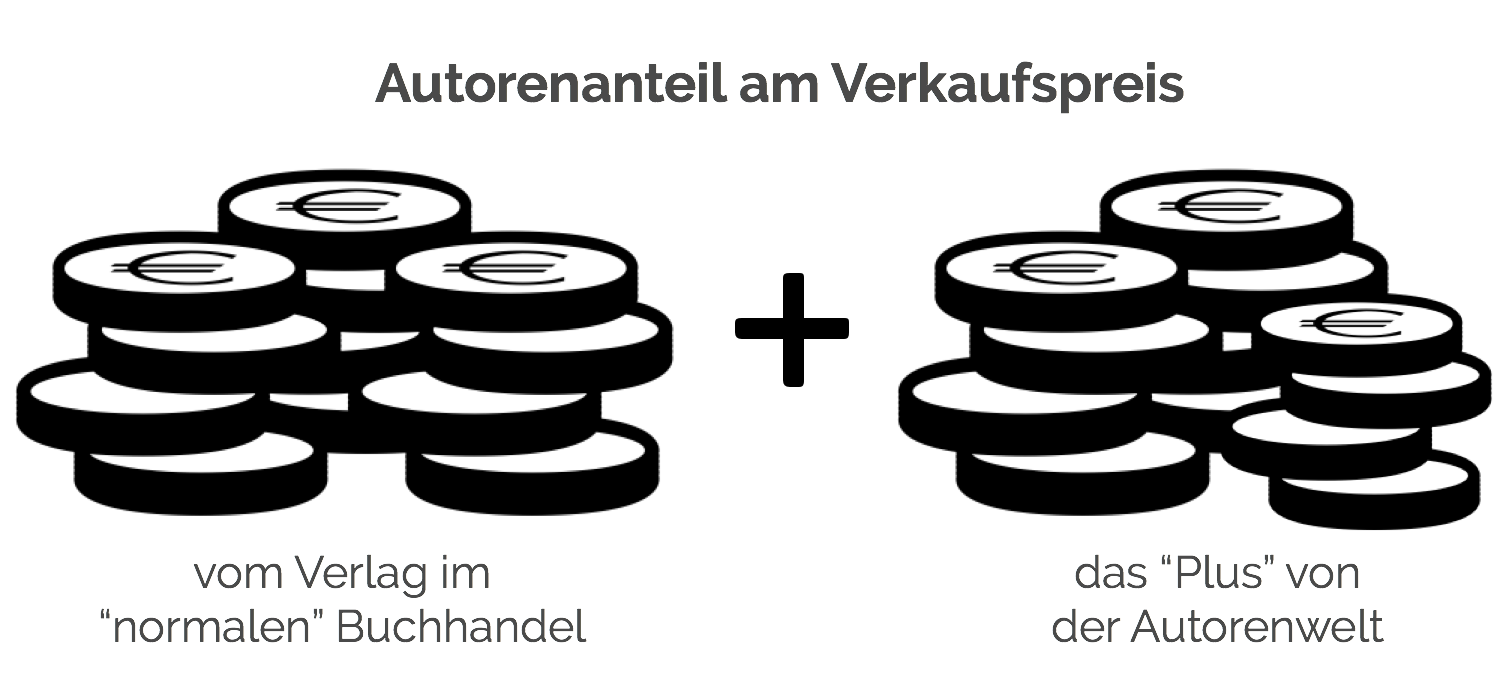
Beschreibung
In my professional career as a pharmaceutical scientist, I have been involved with severalaspectsofthe drugdevelopmentprocessfrompre-INDto commercialization and, somehow, I usually found myself coming back to a stability-related issue. The stability area seemed to draw my utmost interest because in my day-to-day work, my opportunities involved more than one product, and none of the issues were the same.Eachsituationposedchallengesthatusuallyrequiredanexerciseofjudgment, an understandingof regulations,a knowledgeof science, a graspof compliance,and an appreciation of common practices. Sinceearly2000,Ihavealsobeeninvolvedwithseveraltrainingopportunitiesand I struggled to ?nd good, concise, practical resources, one of which I could just hand to a new scientist who wishes to gain a greater understanding of stability sciences. In addition, I encountered the same questions posted over and over on different stability best practices discussion forums. As a book lover, I also have a good collection of technical books. Unfor- nately, most of the stability related volumes are outdated. Many of these materials are theoretical and do not contain much practical information. I understand that the pharmaceutical industry during this period is quite volatile, and guidelines are changingrapidlywhileregulatoryagenciesareworkingcloselywiththepharmac- tical industryto accommodatethese changes;however,thefundamentalinformation continues to remain quite the same, just as current Good Manufacturing Practices (cGMP) continue to be the standard industry practice. Therefore, I hoped to ass- ble a practical handbook to ?ll this void.
Regulations, Methodologies, and Best Practices
Regulations, Methodologies, and Best Practices
Details
| Verlag | Springer US |
| Ersterscheinung | 23. November 2010 |
| Maße | 23.5 cm x 15.5 cm |
| Gewicht | 616 Gramm |
| Format | Softcover |
| ISBN-13 | 9781441927569 |
| Auflage | Softcover reprint of hardcover 1st ed. 2009 |
| Seiten | 389 |


