
Autorenfreundlich Bücher kaufen?!
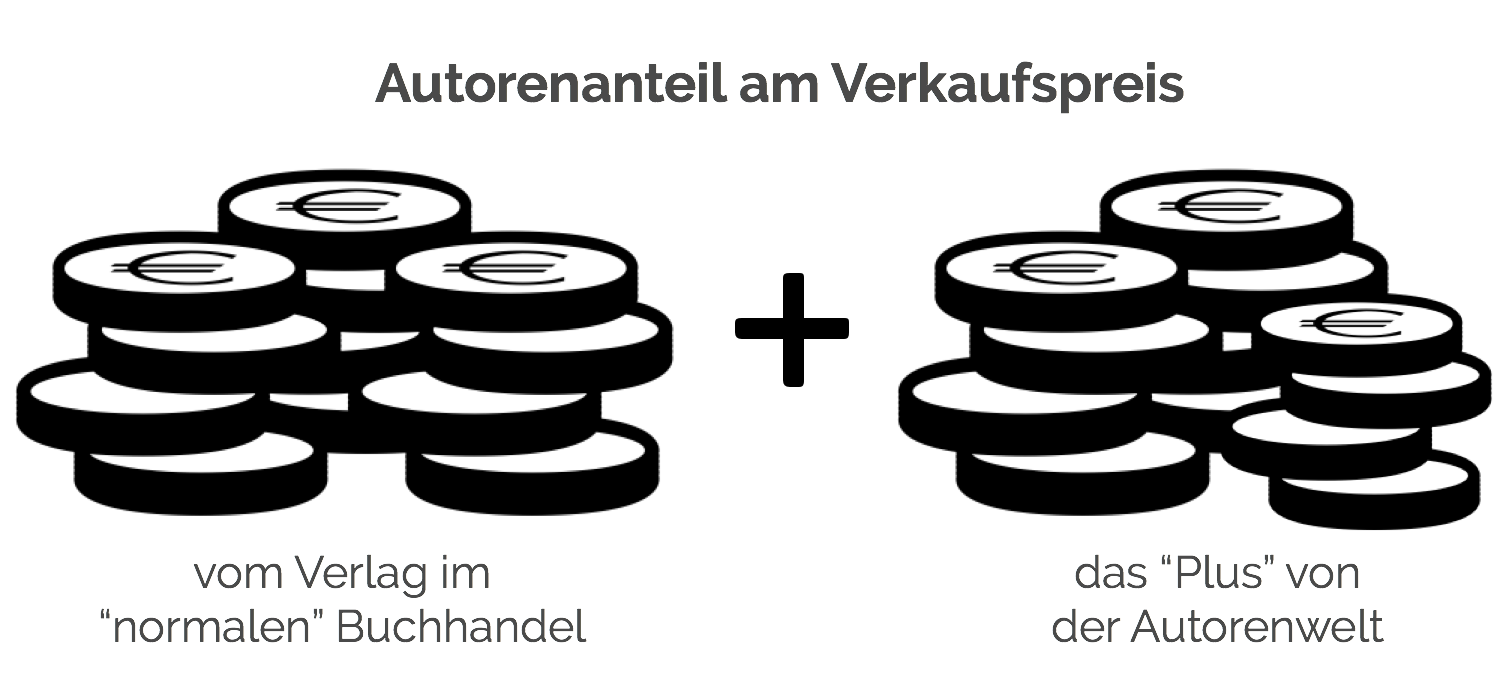
Beschreibung
We developed methods for determination and validation of carvedilol in bulk and pharmaceutical dosage forms by RP- HPLC method with some improvements than the existing method.The analytical procedure described for assay was specific, linear, precise, accurate, and system suitable for determination of carvedilol in bulk and pharmaceutical dosage forms.The observations of the validation parameters such as accuracy, precision, specificity, linearity, shows that the developed methods can be employed for routine analysis of bulk and tablets form of carvedilol. The result obtained from the validation parameters met the ICH and USP requirement.
FOR CARVEDILOL TABLETS BY RP-HPLC
Details
| Verlag | LAP LAMBERT Academic Publishing |
| Ersterscheinung | 19. Mai 2023 |
| Maße | 22 cm x 15 cm x 0.6 cm |
| Gewicht | 131 Gramm |
| Format | Softcover |
| ISBN-13 | 9786206165163 |
| Seiten | 76 |